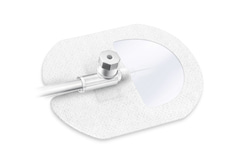
Cosmopor I.V. transparentní jsou neaktivní, jednorázové, samolepicí, sterilní pomůcky
Cosmopor I.V. transparent
- Produkty
- Použití
- Multimedia
Produkty

Cosmopor I.V. transparentní jsou neaktivní, jednorázové, samolepicí, sterilní pomůcky skládající se z potaženého laminátu podkladového materiálu s prostupem (potaženým PU fólií, aby bylo možné pozorovat kanyly/katétry během aplikace obvazu) a I.V. otvor pro zavedení kanyly, chráněný silikonovým papírem a včetně samolepicích fixačních proužků připevněných k silikonovému papíru, pro sterilní krytí a fixaci kanyl/katétrů.
Cosmopor I.V. transparent
Cosmopor I.V. transparentní jsou neaktivní, jednorázové, samolepicí, sterilní pomůcky skládající se z potaženého laminátu podkladového materiálu s prostupem (potaženým PU fólií, aby bylo možné pozorovat kanyly/katétry během aplikace obvazu) a I.V. otvor pro zavedení kanyly, chráněný silikonovým papírem a včetně samolepicích fixačních proužků připevněných k silikonovému papíru, pro sterilní krytí a fixaci kanyl/katétrů.
sterilní, individuálně baleno
- Informace o produktech 6 x 5 cm
-
- číslo výrobku
- 9008171
- Obsah
- 1 balení á 100 ks
- EAN
- 4052199501079
- Obsah
- 1 karton á 12 balení
- EAN
- 4052199501086
- Informace o produktech 9 x 7 cm
-
- číslo výrobku
- 9008181
- Obsah
- 1 balení á 100 ks
- EAN
- 4052199501109
- Obsah
- 1 karton á 12 balení
- EAN
- 4052199501116
- Informace o produktech 12 x 10 cm
-
- číslo výrobku
- 9008191
- Obsah
- 1 balení á 50 ks
- EAN
- 4052199501130
- Obsah
- 1 karton á 12 balení
- EAN
- 4052199501147
| Informace o produktech | číslo výrobku | Obsah | EAN | ||
| sterilní, individuálně baleno | |||||
| 6 x 5 cm |
|
1 balení á 100 ks 1 karton á 12 balení | 4052199501079 4052199501086 | ||
| 9 x 7 cm |
|
1 balení á 100 ks 1 karton á 12 balení | 4052199501109 4052199501116 | ||
| 12 x 10 cm |
|
1 balení á 50 ks 1 karton á 12 balení | 4052199501130 4052199501147 | ||
Použití
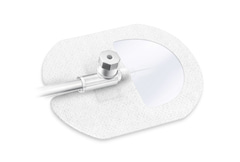
Odstraňte fixační proužky z ochranného krytu číslo 1 a použijte je k fixaci katétru, křidélek kanyly, jehly kanyly a/nebo hadiček. Sejmutí a aplikaci fixačních proužků lze provést i po kompletní aplikaci přípravku Cosmopor I.V. transparentní pro další fixaci kanyly/křídel kanyly a samotného obvazu (viz krok 4). V takovém případě ochranný kryt číslo 1 zlikvidujte až po odstranění a nasazení fixačních proužků.
Po odstranění ochranného krytu číslo 1 aplikujte obvaz s průhledným okénkem vycentrovaným na místo zavedení/výstupu kanyly. Upozorňujeme, že v případě Cosmopor I.V. transparentní 12x10cm, horní stranu ochranného krytu číslo 3 lze použít jako přídavný uchopovací bod pro přesné umístění obvazu.
Uvolněte ochranný kryt číslo 2 z nožiček obvazu a přiložte je na katétr, kanylu a/nebo křídla kanyly. Jemně přitlačte celý obvaz na kůži, přizpůsobte jej konturám těla a tvaru kanyly nebo katetru pro bezpečnou fixaci.
Pro Cosmopor I.V. transparentní 12x10cm, poté opatrně sejměte zbývající ochranný kryt číslo 3 a poté přiložte na pokožku horní část obvazu.
Aplikace fixačních proužků může být také provedena po kroku číslo 3 pro další fixaci katétru, kanyly, křídel kanyly a/nebo hadiček (pokud jsou použitelné) a samotného obvazu. Odstranění Chcete-li bezpečně odstranit nebo vyměnit Cosmopor I.V. transparentní po individuální době aplikace, opatrně odstraňte fixační proužky, poté povolte okraj nožiček obvazu a zcela je sejměte. Po odstranění nohou je třeba kanylu opatrně přitlačit k místu vpichu, aby se jehla kanyly nepohybovala. Poté musí být zbytek obvazu natažen a odstraněn celý, aby se zabránilo pohybu kanyly a jehly.